Calcium Carbide is a man mad compound with the chemical formula CAC^2 and has a molar mass of 64.009g/mol. Calcium carbide is used for steel making, acetylene production, spelunking and mining. For mining, calcium carbide is used for the miners' lamps.
Structure
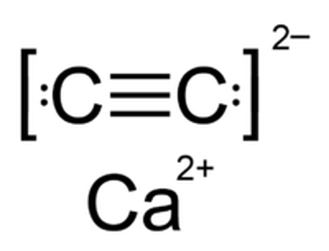
Calcium Carbide is formed by a triple molecular bound between calcium and carbon. It's structure takes makes up 88% of it's mass.
This site was created by Regan Cole
Chemical Compound Project Website
Chemical Compound Project Website
